What "growing a tooth" actually means
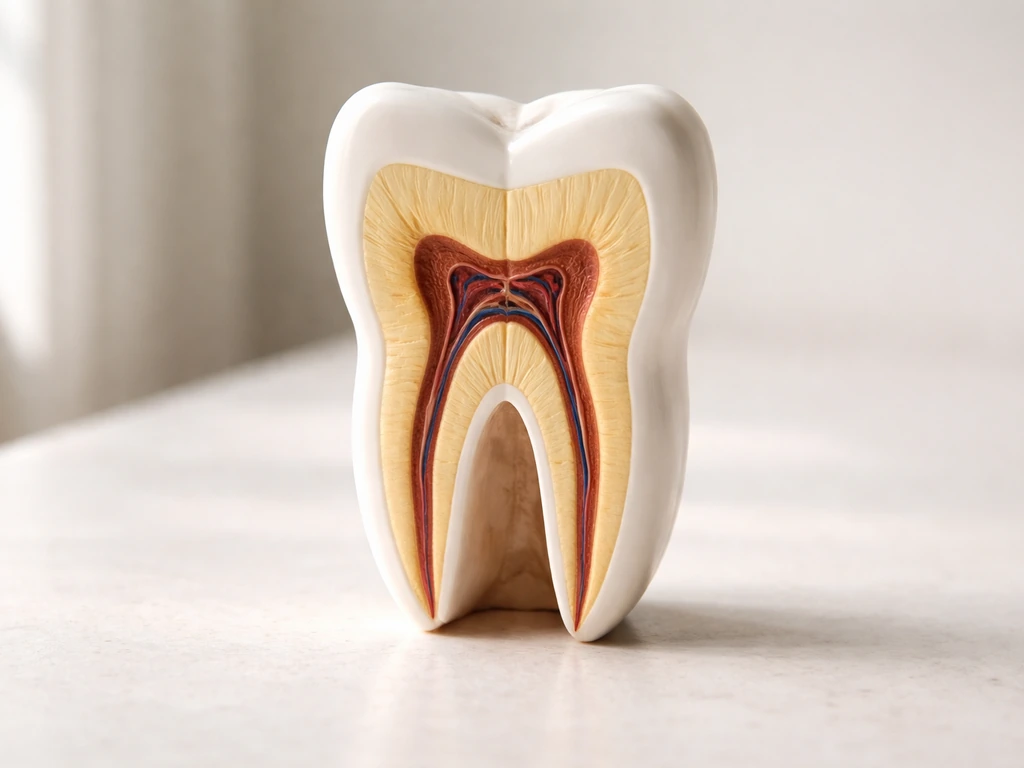
When people ask whether stem cells can grow teeth, they usually have one of several very different things in mind. A real tooth is not one simple structure. It is made up of at least four distinct tissues, and regenerating any one of them is a completely different biological challenge from regenerating another.
- Enamel: the hard outer shell, the most mineralized tissue in the human body. Enamel-forming cells (ameloblasts) are lost after a tooth erupts, which is why enamel cannot repair itself once damaged.
- Dentin: the calcified layer beneath enamel. Odontoblasts (the cells that make dentin) do persist in the pulp, so limited dentin repair is biologically possible and has been demonstrated in research settings.
- Pulp: the soft living core containing nerves and blood vessels. This is the target of most current stem-cell dental research.
- Periodontal ligament and cementum: the anchoring system that connects a tooth root to the jawbone. Without this, a bioengineered tooth would have no functional attachment, no sensation, and no shock absorption.
Growing a complete functional tooth means producing all of these tissues, integrating them with jaw bone, establishing blood supply, restoring nerve connections, and anchoring the whole structure with a functioning ligament. That is a long list of biological steps, and researchers have not yet accomplished all of them together in humans. Most current stem-cell dental work targets one piece of that puzzle, usually the pulp-dentin complex inside an existing tooth, rather than generating an entirely new tooth from scratch.
What the evidence actually shows right now
The most clinically advanced stem-cell dental work involves dental pulp stem cells (DPSCs) and mesenchymal stem cells (MSCs) being used inside existing teeth that have been damaged or had root canals. A 2024 review of MSC-based approaches in human dental clinical trials was clear on this point: current clinical approaches do not result in the regeneration of a functional dentin-pulp complex. The researchers recommended using the phrase "guided endodontic repair" rather than "full pulp-dentin regeneration," because what is actually happening is repair and partial tissue formation, not true biological recreation of the original structure.
A separate 2024 review in PubMed found that while preclinical research (mostly in animals) has advanced considerably, the clinical translation in humans still supports tissue repair rather than reliably rebuilding a complete, functional dentin-pulp unit. The main hurdles are getting blood vessels to grow into the new tissue (angiogenesis), restoring nerve connections (innervation), and making sure the new tissue integrates properly with existing tooth structure. Each of those is an active, unsolved research problem.
For context on how unusual regeneration-capable biology can get, it is worth noting that some of the most striking "dental" tissue growth happens not in normal teeth but in unusual contexts. Bodies can produce tooth-like calcified structures in unexpected places, as anyone who has read about whether cysts can grow teeth already knows. The biology that allows tooth-like tissue to form in odd locations is actually one of the things researchers study to understand how tooth formation could be redirected therapeutically.
How stem-cell tooth research is actually done
Tooth organoids and bioengineering approaches

In research labs, scientists grow "tooth organoids," which are tiny three-dimensional structures that mimic the cellular organization of a developing tooth. These are created by combining dental epithelial cells and mesenchymal stem cells and allowing them to self-organize, mimicking the embryonic signaling cascades that produce teeth during fetal development. In mice and other animal models, fully formed teeth have been grown from these kinds of cell combinations and implanted into jaw bone with partial functional success.
Bioprinting is another approach being explored: using 3D printers loaded with stem-cell-containing bioink to build scaffolds that approximate tooth geometry, then encouraging the cells to differentiate into the appropriate tissue types. The resulting structures are getting closer to real tooth architecture, but getting nerves and blood vessels to grow in remains the biggest challenge. A printed scaffold that lacks innervation will not give you sensation or proprioception, the ability to feel how hard you are biting, which is a basic function of a natural tooth.
Where animals are ahead of humans
Many animals can regenerate teeth naturally throughout their lives: sharks, crocodiles, and some fish replace teeth continuously. Research into why those animals retain this ability and humans do not has been illuminating. It turns out the genetic and signaling pathways for tooth development are not fully switched off in adult humans. There is active research into whether those pathways can be reactivated. Japanese researchers, for instance, have been working on a drug-based approach to stimulate dormant tooth-forming cells, with early clinical trials in humans beginning recently. But those trials are targeting people with rare genetic conditions causing missing teeth, not the general population with standard tooth loss.
The teratoma connection

One of the more striking demonstrations of stem-cell-driven tooth tissue formation happens naturally, if accidentally, in teratomas. These tumors arise from pluripotent cells and can spontaneously produce recognizable dental tissues including enamel and dentin. If you are curious about the biology behind this, reading about why teratomas grow teeth gives you a vivid real-world example of just how powerful undirected pluripotent stem cell differentiation can be, and why controlling that power for therapeutic purposes is so complex.
Who could realistically benefit, and when
The honest answer here depends heavily on your specific situation, your age, and what type of tooth problem you have.
| Patient Situation | Stem-Cell Option Today | Realistic Timeline for More |
|---|
| Child with damaged baby tooth | Pulp repair/capping with biologic materials (not true stem-cell therapy) | N/A — baby tooth will be replaced naturally |
| Child with permanent tooth damage (immature root) | Regenerative endodontics (partially stem-cell based) in clinical use | Already available in select cases, ask an endodontist |
| Adult with damaged pulp in existing tooth | Guided endodontic repair in trials — not standard care yet | Possibly 5-10 years for broader availability |
| Adult with missing tooth (implant candidate) | No stem-cell replacement available — implant is current best option | Full tooth regeneration: 10-20+ years minimum estimate |
| Person with genetic condition causing missing teeth | Active early-phase clinical trials (drug-based pathway activation) | Early trials ongoing; not widely available |
Regenerative endodontics is the area where stem-cell principles have already made it closest to clinical use. In young patients with permanent teeth that have immature (not fully formed) roots and damaged pulp, a technique using the patient's own blood clot as a scaffold to encourage stem cells already present in the tooth socket has been used to allow continued root development. This is not exactly stem-cell therapy in the lab-science sense, but it uses the same biological principles and is recognized by endodontic professional bodies as a legitimate treatment in specific cases.
Adults with fully formed teeth and missing teeth have the fewest options right now. It is also worth noting that some oral anatomy questions involve bone and tissue changes that are not tooth regeneration at all. For example, readers sometimes ask about bony growths in the mouth that develop over time, and understanding whether mandibular tori can grow is a separate topic but one that illustrates how jaw tissues can undergo structural changes in adulthood, which is relevant background for understanding bone integration challenges in tooth implantation research.
Risks, ethics, and the cost problem
Unproven clinics are a real danger
This is the section that matters most if you are actively shopping for a stem-cell dental treatment. The International Society for Stem Cell Research (ISSCR) 2024 guidelines explicitly condemn administering unproven stem-cell interventions outside of properly structured clinical research. The guidelines specifically call out premature commercialization and inaccurate marketing that claims or implies stem-cell action without proven safety or efficacy. In plain terms: clinics that advertise stem-cell tooth regrowth as a service available today, outside of a registered clinical trial, are selling something that does not have an established evidence base.
The risks of these unregulated procedures are not trivial. Injecting or implanting stem-cell preparations into oral tissues can cause infections, unwanted tissue growth, immune reactions, and in cases involving pluripotent stem cells, there is a theoretical risk of tumor formation. This is not science fiction; it is a documented concern with uncontrolled pluripotent cell use. The same cellular power that makes teratomas produce tooth-like structures (and that makes people ask things like whether tumors can grow teeth) is the biological reason that using undirected pluripotent cells in humans carries real safety risks.
Cost and access reality
Even the legitimate experimental work is expensive and inaccessible to most people. Participating in a clinical trial is typically free to the patient (the research institution bears the cost), but trials have strict eligibility criteria and are conducted in limited academic medical centers. Any clinic quoting you a price for a stem-cell tooth treatment outside of a registered trial is almost certainly not offering you something that has been vetted for safety and effectiveness.
There is also a meaningful ethical conversation happening in the research community about who will have access to tooth regeneration therapies if and when they arrive. Early versions of these treatments will almost certainly be expensive and available only in wealthy healthcare systems, raising real questions about equitable access. This is part of why regulatory bodies and research ethics frameworks are paying close attention to how commercialization happens in this space.
The ovarian teratoma context
A related topic that sometimes comes up in conversations about stem cells and unusual tissue growth is the formation of dental structures in reproductive tissues. The biological curiosity behind whether ovaries can grow teeth is directly connected to the same pluripotent stem cell biology that makes tooth regeneration research possible. Understanding those connections helps clarify both the promise and the genuine risk of working with highly potent stem cells.
What to do right now: questions to ask and how to find real trials
Questions to bring to your dentist or specialist
- Am I a candidate for regenerative endodontics? (Ask this especially if you have a young patient with an immature permanent tooth with pulp damage.)
- Are there any registered clinical trials at academic institutions near me that I might qualify for?
- What is your recommendation for my specific case today, given that stem-cell tooth regrowth is not yet a standard clinical option?
- If I have seen a clinic advertising stem-cell tooth regrowth as an available service, can you help me evaluate whether that claim is legitimate?
How to find legitimate clinical trials
In the United States, all registered human clinical trials must be listed at ClinicalTrials.gov. Search for terms like "dental pulp stem cells," "tooth regeneration," or "regenerative endodontics" to find currently enrolling trials. In other countries, look for the equivalent national registry (the WHO has a global registry network). Legitimate trials will have a registered number, a listed principal investigator at an academic institution or hospital, and a detailed protocol explaining what is being tested and how participant safety is protected.
Red flags that suggest a clinic is not running a legitimate trial: no registered trial number, a fee charged to the patient for a "stem-cell treatment," claims that the treatment is proven or FDA-approved when no such approval exists for tooth regrowth, and vague language about what type of cells are being used or where they come from. If a clinic cannot answer specific questions about the source of the stem cells, the regulatory status, and the evidence base, that is a strong signal to walk away.
What your best options are today
For missing teeth, dental implants remain the gold standard in 2026. They integrate with jaw bone, restore full chewing function, and have a 15-plus year track record of safety and effectiveness. For damaged teeth with pulp involvement, modern root canal therapy combined with restoration is highly effective and widely available. For younger patients with specific types of pulp damage, ask an endodontist specifically about regenerative endodontic procedures, which are closer to the stem-cell biology frontier than standard root canals.
Stem-cell tooth regeneration is not science fiction. The research is real, the progress is real, and the path to clinical treatment is becoming clearer. But the honest timeline for a routine clinical option that grows you a complete new tooth is probably still at least a decade away, possibly longer. The people who will benefit from early breakthroughs are most likely going to be patients who qualify for clinical trials at academic research centers, not people paying for commercial treatments at private clinics. Knowing that distinction, and asking the right questions of the right specialists, is the most useful thing you can do with this information right now.